![Determine the structure and magnetic behaviour of [CoCl4 ]²- using valence bond theory. according to board - Brainly.in Determine the structure and magnetic behaviour of [CoCl4 ]²- using valence bond theory. according to board - Brainly.in](https://hi-static.z-dn.net/files/dbc/4e4b8ae040fa0c9d9e6932ef4496ad25.jpg)
Determine the structure and magnetic behaviour of [CoCl4 ]²- using valence bond theory. according to board - Brainly.in
![Consider this reaction: [Co(H2O)6]2+ + 4 Cl- -> ? [CoCl4]2- + 6 H2O The octahedral starting material is pink in color, absorbing visible light with ?max = 510 nm. The tetrahedral product Consider this reaction: [Co(H2O)6]2+ + 4 Cl- -> ? [CoCl4]2- + 6 H2O The octahedral starting material is pink in color, absorbing visible light with ?max = 510 nm. The tetrahedral product](https://homework.study.com/cimages/multimages/16/studycomoctahedraltetrahedraldq10shownd7system1969898108720194826.png)
Consider this reaction: [Co(H2O)6]2+ + 4 Cl- -> ? [CoCl4]2- + 6 H2O The octahedral starting material is pink in color, absorbing visible light with ?max = 510 nm. The tetrahedral product
![Answer in brief. [CoCl4]2- is tetrahedral complex. Draw its box orbital diagram. State which orbitals participate in hybridization. - Chemistry | Shaalaa.com Answer in brief. [CoCl4]2- is tetrahedral complex. Draw its box orbital diagram. State which orbitals participate in hybridization. - Chemistry | Shaalaa.com](https://www.shaalaa.com/images/_4:65f664b9fefa456ba348e04d03e2557a.png)
Answer in brief. [CoCl4]2- is tetrahedral complex. Draw its box orbital diagram. State which orbitals participate in hybridization. - Chemistry | Shaalaa.com
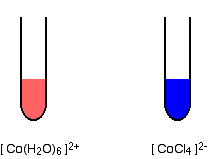
The formation of CoCl4 2- from Co 2+ and Cl- is endothermic. Are the color changes that accompany heating and cooling of equilibrium mixture in accord with Le Chatelier's principle? | Socratic

Describe the distribution of d electrons in the (CoCl_4)^2- ion. The ion has a tetrahedral geometry. Assume a high-spin complex. | Homework.Study.com
![Cobalt tetrachloride [CoCl4]2- ion - Structure - hybridization - color - leChatelier principle - YouTube Cobalt tetrachloride [CoCl4]2- ion - Structure - hybridization - color - leChatelier principle - YouTube](https://i.ytimg.com/vi/TBGsBJMN9fU/sddefault.jpg)
Cobalt tetrachloride [CoCl4]2- ion - Structure - hybridization - color - leChatelier principle - YouTube
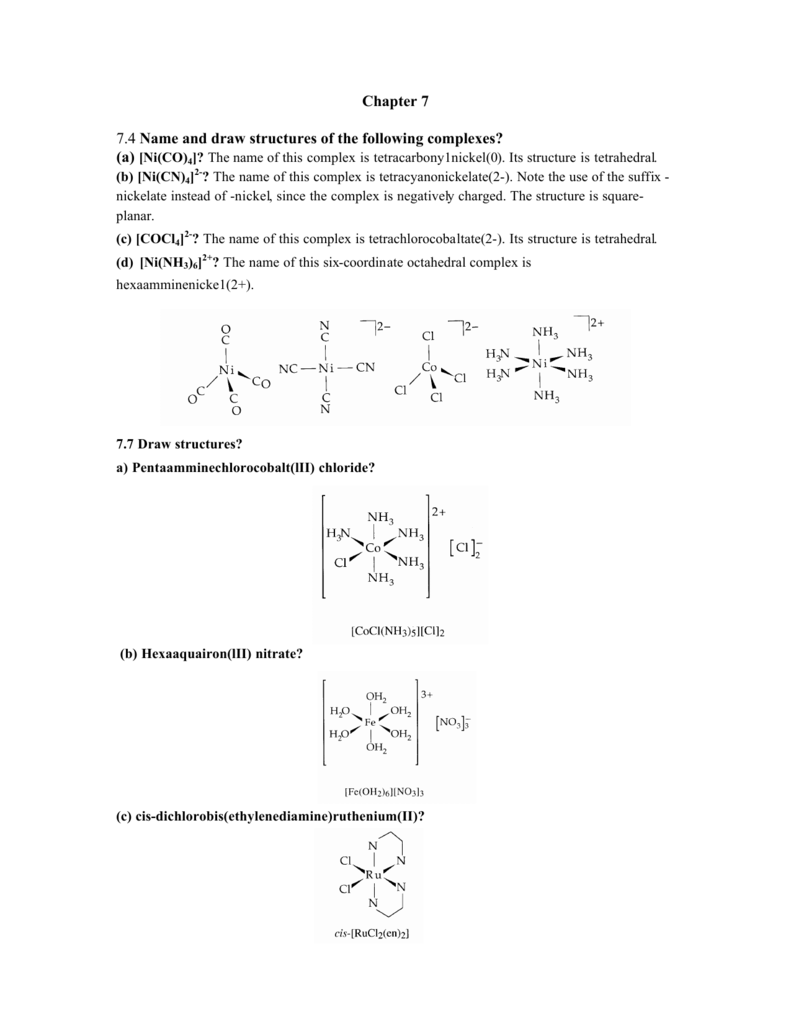
69 The no of geometrical isomers for octahedral [CoCl4(NH3)2 ] , square planar [AuBr2Cl2] , and [PtCl2(en) ] are
![SOLVED: What is the correct formula for sodium tetrachlorocobaltate(I)? a. NaZ[CoCl4] b. Na2[CoCl4] c. Na4[CoCl4] d. Na[CoCl4] e. Na[CoCl4] SOLVED: What is the correct formula for sodium tetrachlorocobaltate(I)? a. NaZ[CoCl4] b. Na2[CoCl4] c. Na4[CoCl4] d. Na[CoCl4] e. Na[CoCl4]](https://cdn.numerade.com/ask_previews/9d18200b-36c3-4d2c-904b-4866aaaff2ba_large.jpg)
SOLVED: What is the correct formula for sodium tetrachlorocobaltate(I)? a. NaZ[CoCl4] b. Na2[CoCl4] c. Na4[CoCl4] d. Na[CoCl4] e. Na[CoCl4]
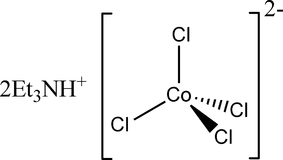
![Hybridization-Ni(CO)4 | [Ni(CN)4]2-| [Ni(Cl)4]2- | Structure-Parmagnetic-Diamagnetic-Examples-dsp2 Hybridization-Ni(CO)4 | [Ni(CN)4]2-| [Ni(Cl)4]2- | Structure-Parmagnetic-Diamagnetic-Examples-dsp2](http://www.adichemistry.com/jee/qb/coordination-chemistry/1/q1-3.png)
![SOLVED: State the nomenclature of (NH4)2[CoCl4] SOLVED: State the nomenclature of (NH4)2[CoCl4]](https://cdn.numerade.com/ask_previews/139d716c-5ff4-455b-a952-0f605374158a_large.jpg)
![Write the IUPAC name and hybridisation of K(2)[CoCl(4)(en)] Write the IUPAC name and hybridisation of K(2)[CoCl(4)(en)]](https://static.doubtnut.com/ss/web/1312549.webp)
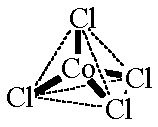
![CoCl4]2- - Cobalt tetrachloride CoCl4]2- - Cobalt tetrachloride](https://www.chemtube3d.com/images/gallery/inorganicsjpgs/cocl42-.jpg)
![Hybridization-Ni(CO)4 | [Ni(CN)4]2-| [Ni(Cl)4]2- | Structure-Parmagnetic-Diamagnetic-Examples-dsp2 Hybridization-Ni(CO)4 | [Ni(CN)4]2-| [Ni(Cl)4]2- | Structure-Parmagnetic-Diamagnetic-Examples-dsp2](http://www.adichemistry.com/jee/qb/coordination-chemistry/1/q1-2.png)

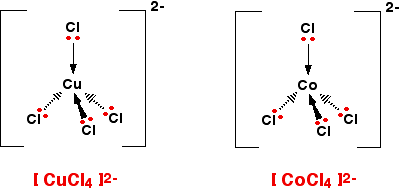

![Solved Question 13 What is the IUPAC name for K2[CoCl4]? A. | Chegg.com Solved Question 13 What is the IUPAC name for K2[CoCl4]? A. | Chegg.com](https://media.cheggcdn.com/study/dd6/dd61b5b6-3c0e-4c85-917b-cea9c36f4256/image.png)
![The CFSE value of tetrahedral [CoCl4]2 complex is: The CFSE value of tetrahedral [CoCl4]2 complex is:](https://df0b18phdhzpx.cloudfront.net/ckeditor_assets/pictures/1444484/original_22.png)
![Predict the hybridisation and geometry of `[CoCl_(4)]^(2-) and [Co(CN)_(4)]^(2-)` Predict the hybridisation and geometry of `[CoCl_(4)]^(2-) and [Co(CN)_(4)]^(2-)`](https://i.ytimg.com/vi/m7yduxa5Ysw/maxresdefault.jpg?sqp=-oaymwEmCIAKENAF8quKqQMa8AEB-AH-CYAC0AWKAgwIABABGGUgZShlMA8=&rs=AOn4CLDioezo6yafUtFRdVQZgUlgZXiOWg)